  
There are limitations to this method that are balanced by the advantages. Comparison to current methods to produce hmAbs These humanizing techniques represent a significant outlay of time and resources 9. Finally, for potential therapeutic applications, the Fab that is produced by phage display libraries or in other species (mice) must be cloned and fused to a human Fc backbone and expressed in a human cell line. These same applications have been hindered by technologies using immortalized B-cell lines because of the relatively few specific antibodies isolated that can be generated. The mAbs generated by in vitro methods or in other species do not provide a true evaluation of the epitope specificities that humans generate in vivo, limiting the use of these techniques for applications such as epitope discovery and vaccine development or evaluation. Although phage display technology uses fully human heavy and light chain variable genes, the heavy and light chains are randomly paired in vitro, and so are more likely to induce anaphylactic responses as foreign proteins or to be autoreactive if therapeutic uses are the goal. Current phage display and related platforms spend extensive amounts of time identifying the few candidate antibodies present and a significant portion of these turn out to be of low affinity 9. 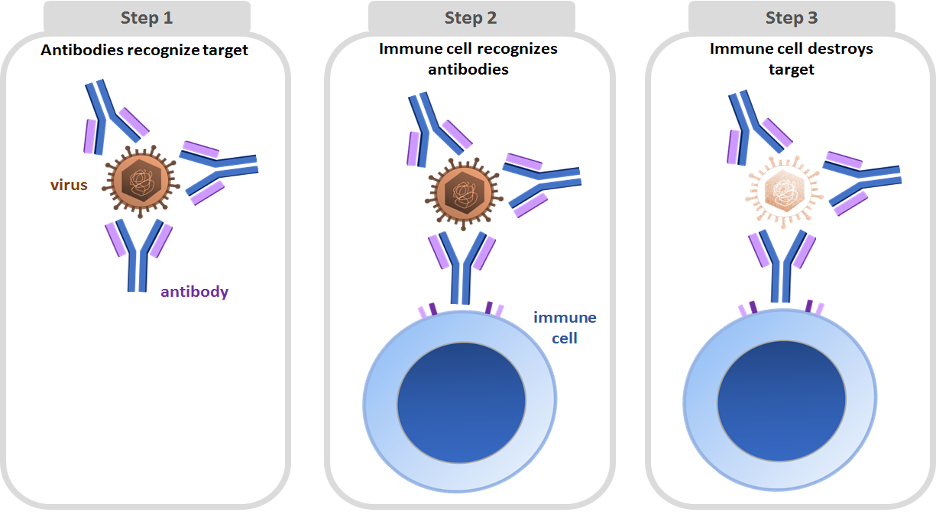
In methods requiring immortalized B-cell lines, the extensive subcloning and overall shotgun approach limit the number of useful antibodies that can be produced even over extensive periods of time 9. However, the technique described herein is more suited for the rapid development of a large library of antibodies with a range of specificities against a particular immunogen. HmAbs can be produced by several methods, including immortalization of B cells with Epstein–Barr virus 2, 3, and the production of B-cell hybridomas 4, humanization of antibodies from other species 5, using phage display libraries 6 or generating antibodies recombinantly from isolated single B cells 7, 8. Because of the wide epitope specificity of the antibodies produced by this method, large numbers of high-affinity antibodies can be produced quickly, yielding panels of diagnostics for rapid antigen screens. 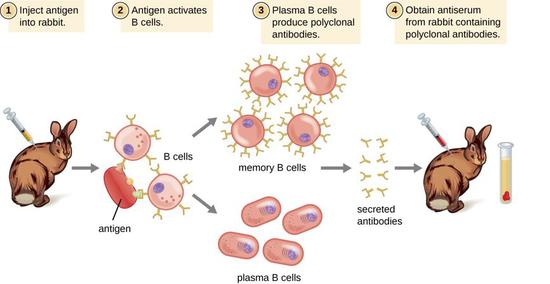
These range from elucidating the interactions of particular antibodies and antigens to exploring basic B-cell immunology or to producing valuable therapeutics. This ability to express and characterize antigen-specific hmAbs is extremely useful for a variety of applications. By this technique, it is possible for a lab experienced with the process to produce milligrams of human monoclonal antibodies (hmAbs) in as little as 28 d. This protocol is derived from strategies developed in our recent study characterizing the human B-cell response to influenza 1. Although dependent on having an ongoing immune response, the approach described herein can be used to rapidly generate numerous antigen-specific hmAbs in a short time. Although previous methodologies to produce hmAbs, including B-cell immortalization or phage display, can be used to isolate the rare specific antibody even years after immunization, in comparison, these approaches are inefficient, resulting in few relevant antibodies. This protocol can be completed with as little as 20 ml of human blood and in as little as 28 d when optimal. This method uses established techniques but is novel in their combination and application. The expressed antibodies can then be purified and assayed for binding and neutralization. The antibody genes of the ASCs are then amplified by RT-PCR and nested PCR, cloned into expression vectors and transfected into a human cell line. Antibody-secreting cells (ASCs) are isolated from whole blood collected 7 d after vaccination and sorted by flow cytometry into single cell plates. We describe herein a protocol for the production of antigen-specific human monoclonal antibodies (hmAbs). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
